Gastric Balloon Obesity Treatment Deaths May Encourage Lawsuits
Get Legal Help Today
Find the right lawyer for your legal issue.
Secured with SHA-256 Encryption
UPDATED: Jul 16, 2021
It’s all about you. We want to help you make the right legal decisions.
We strive to help you make confident insurance and legal decisions. Finding trusted and reliable insurance quotes and legal advice should be easy. This doesn’t influence our content. Our opinions are our own.
Editorial Guidelines: We are a free online resource for anyone interested in learning more about legal topics and insurance. Our goal is to be an objective, third-party resource for everything legal and insurance related. We update our site regularly, and all content is reviewed by experts.
UPDATED: Jul 16, 2021
It’s all about you. We want to help you make the right legal decisions.
We strive to help you make confident insurance and legal decisions. Finding trusted and reliable insurance quotes and legal advice should be easy. This doesn’t influence our content. Our opinions are our own.
On This Page
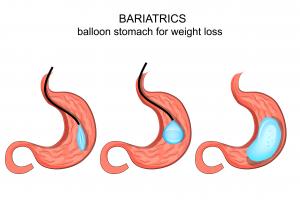
Balloons Used to Treat Obesity
The systems involve the insertion of one or two balloons into the patient’s stomach. The balloons are inserted through the patient’s mouth with an endoscope while the patient is mildly sedated. The balloon is then filled with a saline solution (or some other liquid) until it reaches the size of a grapefruit. The inflated balloon remains in the patient’s stomach for up to six months. During those months, the patient is encouraged to exercise and eat a healthy diet.
The gastric balloon is intended to take up space in the stomach, leaving less room for food. Since (as overeaters are well aware) the stomach expands to accommodate food intake, it is not clear why gastric balloon treatment works. The Food and Drug Administration (FDA) theorizes that the balloon leaves patients feeling full and therefore quells the appetite. However, the FDA notes that the mechanism leading to weight loss is not fully understood.
A clinical trial offered in support of the dual balloon system compared recipients of the treatment to study participants who underwent a similar procedure in which no balloons were inserted. The participants who received the balloons lost about 14 pounds, while the participants who did not receive balloons lost about 7 pounds.
Losing weight, of course, does not assure that the patient will not regain the weight after the treatment ends. The cost of gastric balloon treatment averages $8,150 and is not typically covered by health insurance. Members of a panel that gives advice about Medicare coverage decisions recently expressed skepticism about the medical value of gastric balloon treatment.
FDA Safety Alert
The FDA alerted healthcare providers to five patient deaths since 2016 that followed soon after the insertion of a gastric balloon. The FDA’s letter states that all five deaths occurred within a month of balloon placement. In three cases, death occurred within three days after balloon placement.
The letter emphasizes that the FDA has not determined the “root causes” of the patient deaths and therefore could not attribute the deaths to the device itself or to its insertion. The regulatory agency recommended that doctors closely monitor patients who have receive a gastric balloon implant for potential complications.
The letter noted that two other patient deaths that occurred in the same time period resulted from “complications associated with balloon treatment” — a gastric perforation in one case and an esophageal perforation in the other. The FDA is studying other potential complications, including acute pancreatitis and spontaneous over-inflation of the balloons.
Legal Liability
It may be that the deaths were caused by a failure to insert the balloons properly. Patient deaths that are caused by insertion of the balloon may expose the doctor who performed the procedure to a medical malpractice lawsuit.
State laws vary, but the general rule is that doctors must provide patients with the standard of care that a prudent doctor who practices in the same field of medicine would provide. Doctors who cause an injury because they failed to provide that standard of care can generally be held accountable for their negligence in a medical malpractice lawsuit.
If the devices themselves cause a death, it may be possible to hold the manufacturer of the device accountable in a product liability lawsuit. State laws again vary, but injury victims usually need to prove the existence of a defect in the design or manufacture of the product. In some cases, proving the defect establishes liability and no further proof of negligence is required.
Proving that a medical device is defective, however, can be very difficult. The manufacturers of the gastric balloon treatment systems obtained FDA approval of their devices by presenting studies to demonstrate that the treatment is safe. The fact that death quickly followed treatment in a handful of cases is not usually sufficient proof to overcome the manufacturers’ product safety studies. Rather, product liability lawyers generally need to provide expert evidence to establish that the death was caused by a specific design or manufacturing defect in the gastric balloon system.
At this point, it is unclear whether any evidence will be marshalled to establish that the balloon devices are defective, or whether patient deaths can be attributed to medical malpractice. However, the fact that patients are dying after gastric balloon treatments, and that the deaths caused the FDA to caution doctors about a potential problem, raises the possibility that product liability and medical malpractice lawyers may soon be focusing on deaths that follow gastric balloon obesity treatments.
Get Legal Help Today
Find the right lawyer for your legal issue.
Secured with SHA-256 Encryption
Editorial Guidelines: We are a free online resource for anyone interested in learning more about legal topics and insurance. Our goal is to be an objective, third-party resource for everything legal and insurance related. We update our site regularly, and all content is reviewed by experts.
